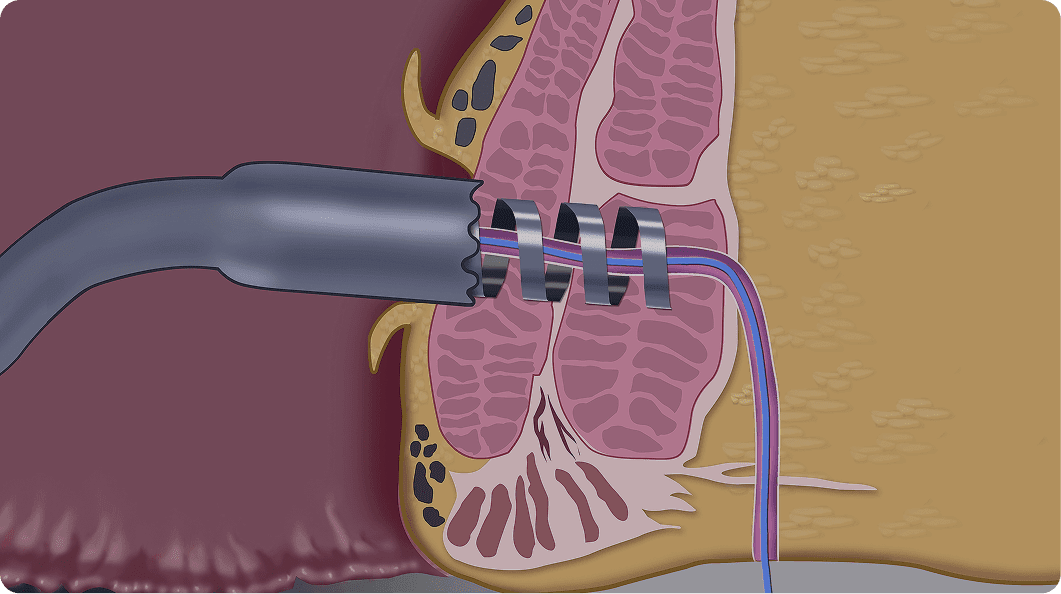
If you’re frustrated by prolonged healing, risk of incontinence, and high recurrence rates, you’re not alone. At Signum Surgical, we believe patients deserve better. Thats why we’ve developed a solution designed to optimize the procedure, promote healing, and prevent recurrence – all in a single procedure.

Patients endure a poor quality of life, experiencing pain, suffering and discharge from the tract and related abscesses.
Current treatment options have poor results, with the risk of causing permanent incontinence, inadequate healing, & repeat surgeries.
There are 90,000 annual surgical procedures performed to treat anal fistula in the U.S. alone.
Please get in touch using the form or reach out directly via the contact details provided.
"*" indicates required fields